Exome era. When a consortium disappears, where does it go and what does it leave behind? I realized last week that exactly 10 years ago, the EuroEPINOMICS Rare Epilepsy Syndrome (RES) consortium pushed the button for the second round of trio exome sequencing, a pivotal event in the history of epilepsy genetics that led to the discovery of more than a dozen genes for developmental and epileptic encephalopathies. The fact that this critical juncture in the history of epilepsy gene discovery went largely unnoticed lies within the nature of research consortia – they form, they work, and they disperse. However, as EuroEPINOMICS was formative for me as a junior researcher, I wanted to dedicate this blog post to the research consortia of the early 2010s and the golden era of epilepsy gene discovery, when I was a EuroEPINOMICS kid.
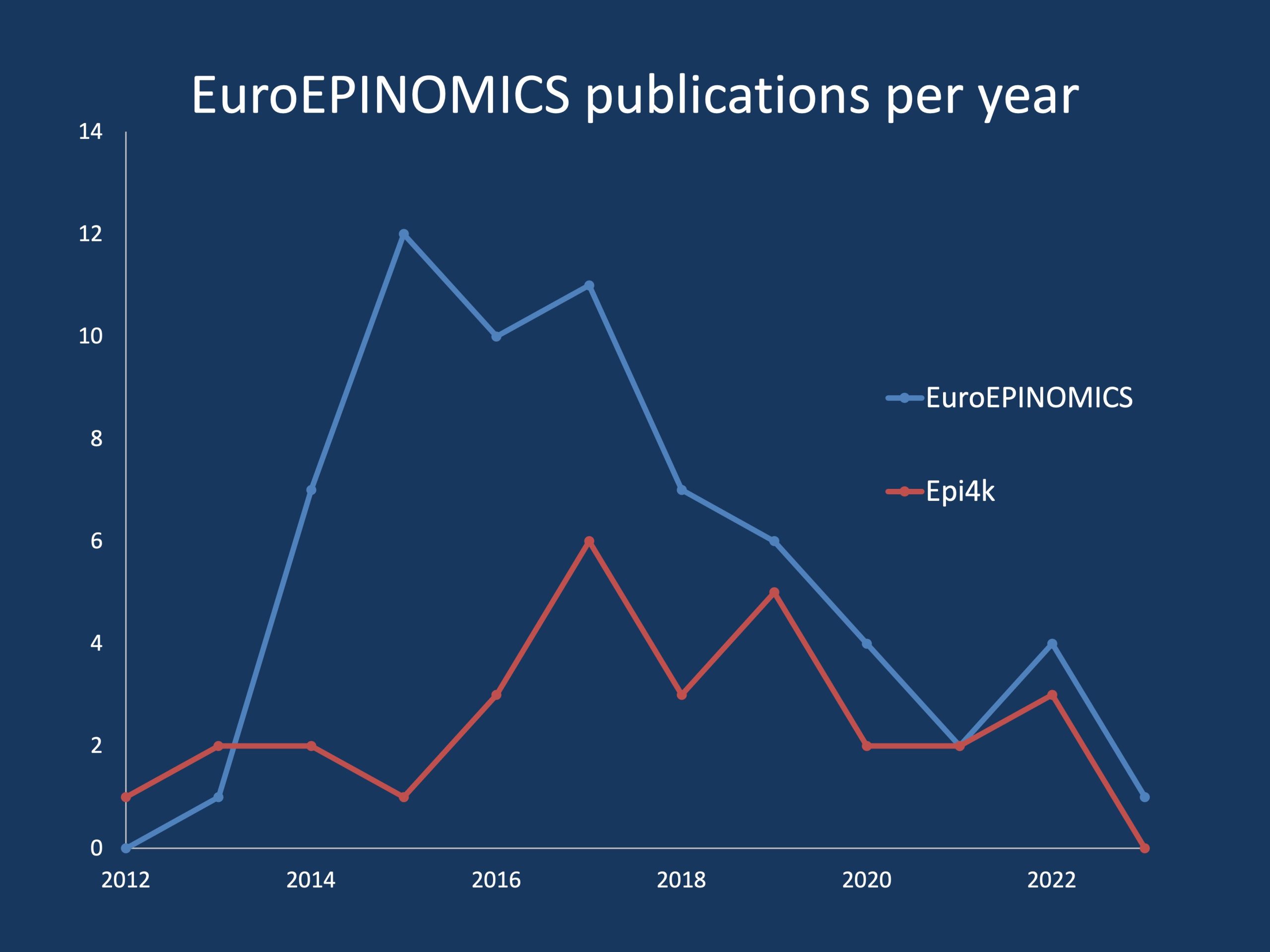
Figure 1. Comparison of the Pubmed-listed publications for the EuroEPINOMICS and Epi4K consortium, the two large international consortia in the 2010s involved in epilepsy gene discovery. The direct comparison of publication numbers is somewhat misleading as Epi4K started out their publication history with a historical Nature paper in 2013. However, over the course of the subsequent decade, EuroEPINOMICS produced twice as many publications, which is largely due to it de-centralized nature of this consortium that gave responsibility of single gene discoveries to individual research groups based on a jointly managed data resources.
History of EuroEPINOMICS. There is no single place where the history of research consortia receives adequate attention, even though the lessons learned in collaborative science are important and highly informative. Sam Berkovic and Peter De Jonghe summarized the experience of the joint Epi4K-EuroEPINOMICS collaboration in a blog post in 2017, but some of the key events of EuroEPINOMICS are at risk of being lost to history. Therefore, I thought it might be timely to revisit our experience a decade later, looking back across the pond. Here are some key events and take away messages that I learned as one of the leaders in EuroEPINOMICS. I would like to ask for forgiveness as I write from my own perspective in this blog post and want to emphasize that EuroEPINOMICS was a consortium supported by a large number of individual investigators in Europe.
ESF. EuroEPINOMICS emerged as European collaborative research project through a funding mechanism of the European Science Foundation (ESF), a funding mechanism that was both unusual and innovative, which no longer exists in this paritcular format. Even though centralized EU funding typically comes to mind when people think about European collaborations, the ESF mechanism worked differently. In brief, it was an agreement between the national funding organization coordinated by the ESF. After a centralized review, the national funding organizations committed to funding the respective projects out of their own budgets. In addition, a subset of the funding was dedicated to collaborative and training efforts. This blog actually emerged out of some of the collaborative efforts supported by the ESF, as did the training events that forged together an entire generation of young European epilepsy genetics researchers.
RES. Within the various EuroEPINOMICS projects, I was part of the leadership of the Rare Epilepsy Syndromes (RES) collaborative, one of the three subprojects of the EuroEPINOMICS initiative. When we initially proposed RES, we weren’t quite sure what would happen. Remember, these were the early days of next generation sequencing and we collaborated with the Sanger Institute to have exome sequencing performed, first on a small subset of 33 patient-parent trios, followed by a second round of 102 trios in 2013. During this time, I learned one of the main lessons in epilepsy genetics that I am still trying to follow: never pay for sequencing.
Never pay for genomes. While this sounds odd, please let me provide you with some detail. This maxim is not about saving money or cheating your way into sequencing projects, but about a single strategic consideration, namely investing in biobanking and personnel rather than raw sequencing costs. This is critical, because despite the cost of sequencing, the availability of biosamples is the limiting factor in rare disease research. It is not the ability to use the latest, flashiest technology, but to have biosamples in hand. This issue persists to this very day, but the emphasis has shifted. Today, this is no longer an issue for developmental and epileptic encephalopathies, but the bottleneck now pertains to conditions such as acquired brain lesions or pediatric migraine, or the availability of plasma or CSF biosamples in rare pediatric neurological disorders.
Leadership. Think of EuroEPINOMICS as a collaborative with a lack of externally reinforced structures. What do I mean by this? Given that the ESF funding mechanism handed the management of grants back to the national funding organizations, there was not a single organization that reviewed and granted the funding. However, the EuroEPINOMICS leadership was in charge of coordinating exome sequencing with the Sanger Institute. This allowed for an interesting and unique “job title” to emerge: junior consortium leader, which eventually became my role. Alongside Peter De Jonghe, I was tasked to lead a major research consortium even though I was only midway through my residency. Generally, consortium leadership is reserved for senior researchers, which is by necessity and design. Typically, only senior Principle Investigators can apply for major grants that result in consortia and most training programs do not allow for meaningful research during residency. Not so in the German training system where it is an expectation and requirement of an academic clinician to perform research throughout residency. This, combined with the unusual ESF funding mechanism, resulted in me becoming a co-leader of a major research consortium in my early 30s, a role which provided me with an interesting perspective on the consortium.
Dynamics. My role was to navigate researchers from nine European countries through a collaborative effort, balancing interests and solving conflicts when they arose. I considered this task as equivalent to performing experiments or sequencing exomes. We developed an intricate framework for rotating authorship that weighed individual contributor efforts and infrastructural support. This mechanism made sure that junior investigators, smaller research groups, clinicians, and staff scientists “behind the scenes” received a fair chance of co-authorship. In brief, we were tasked to responsibly manage a joint resource, the Sanger Institute exome sequencing capacity, to make sure that all collaborators were able to contribute jointly. I am still proud of the fact that one of the first trios we sequenced did not come out of the biobanks of larger research groups, but from our collaborators in Romania. My take-away message: do not underestimate the social dynamics in collaborative research. Even though we typically consider research as objective and neutral, the way that collaborators interact can make or break a consortium.
Gene discovery. There was nothing magic or genius about EuroEPINOMICS, it was simply a consortium that was in the right place at the right time. After somewhat stumbling into the co-discovery of CHD2, the number of gene identifications and publications rose quickly with four Nature Genetics articles in subsequent years and 60 publications in total. It was the time when exome sequencing as a method was still young and many of the genes that we now take for granted were unknown, such as KCNA2, STX1B, HCN1, GRIN2A, GRIN2B, or GRIN1. Due to the distributed funding network, EuroEPINOMICS had the right collaborative framework to emphasize individual publications rather than large consortium publications, which allowed us to establish a gene-matching collaboration in the epilepsy field.
Winding down. Many of the mechanisms used in EuroEPINOMICS only ten years ago are now outdated. Finding individuals with causative variants in a specific gene is now the domain of GeneMatcher and next generation sequencing has moved from being a rare resource to being a common commodity. In addition, Epi25 has become the central consortium for large-scale sequencing, analyzing patient cohorts that I would have thought unthinkable only five years ago. Now, phenotype delineation and natural history studies are the new frontier of data-driven epilepsy genetics research. EuroEPINOMICS was officially concluded with a final meeting in 2014 in Helsinki. However, the real wave of publications only followed in the subsequent years, the lessons learned from collaborative research in this unique setting still have an impact today. Therefore, I will always be a EuroEPINOMICS kid.


