Ring Chromosome 20. This is the Epilepsiome page for Ring Chromosome 20, a genetic epilepsy associated typically with childhood onset epilepsy with unique features such as nonconvulsive status epilepticus.
In a nutshell.
Ring chromosome 20 occurs when a copy of a chromosome 20 has formed a ring rather than the typical linear chromosome structure. It is currently poorly understood how ring chromosome 20 results in epilepsy, particularly with unique features such as explosive onset and nonconvulsive status epilepticus.
Below, we’ll review the presentation of ring chromosome 20, proposed mechanisms of disease, and the importance of methods of genetic diagnosis.
Phenotypes | Genetics | Mechanism | Community
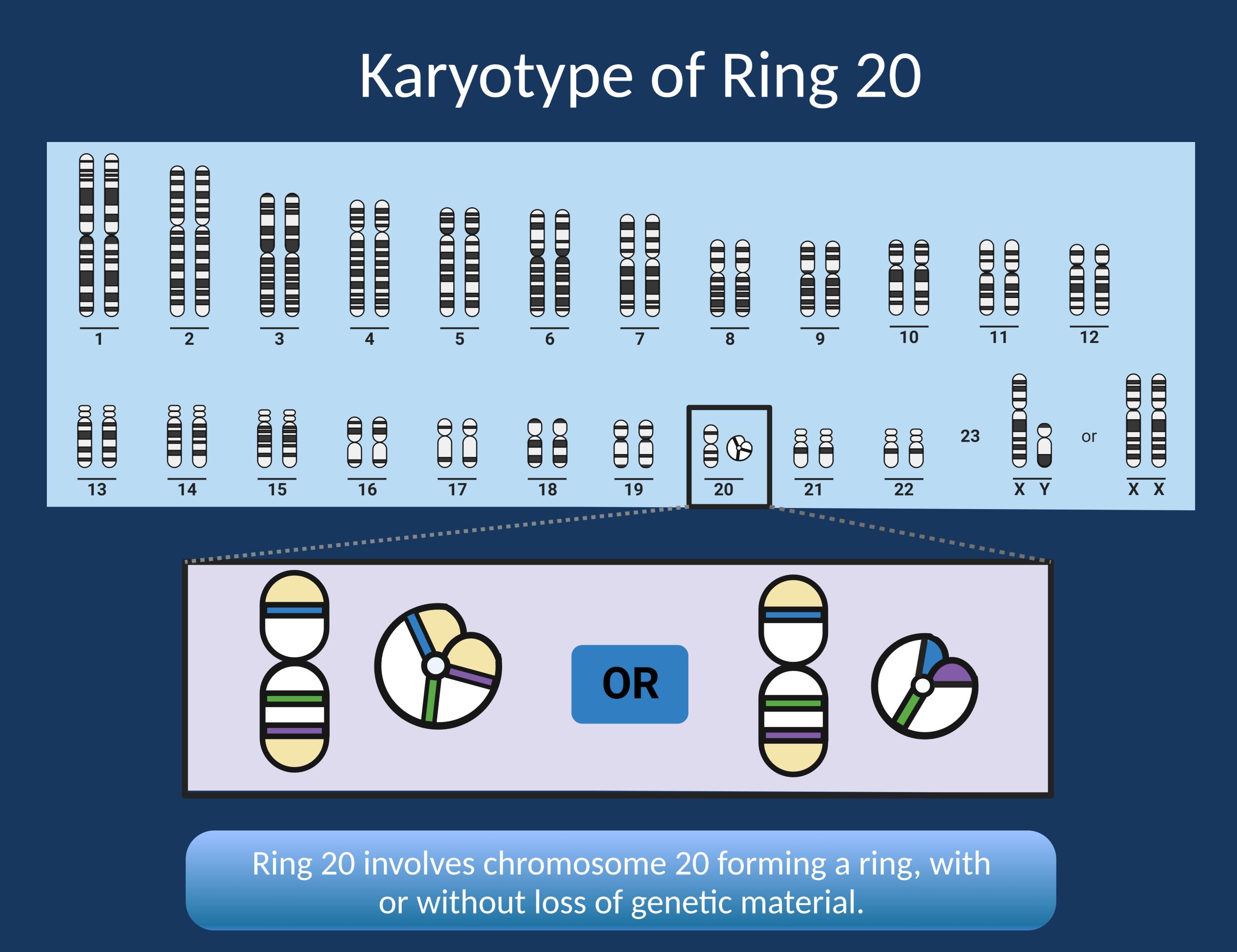
Figure 1. Ring chromosome 20 occurs when chromosome 20 forms a ring structure, with both ends connecting. This can result in no loss of genetic material or loss of genetic material from one or both ends of the chromosome. Karyotype is the only reliable way to make this genetic diagnosis.
Phenotypes
Presentation. Many individuals with ring chromosome 20 do not present to medical attention until early school age (on average, age 7-9 years) after a period of typical development. Initial presentation is typically an explosive onset of epilepsy. Although this is the classic presentation, it is important to consider that individuals can present with earlier or later developmental delays or earlier or later onset of seizures depending on level of mosaicism. Initial presentation is often so sudden and severe that encephalitis or autoimmune encephalopathies are considered as differential diagnoses. Clinical onset of seizures can also include hallucinations, which may be mistaken for psychiatric conditions such as schizophrenia.
Epilepsy. A triad of epilepsy features seen in ring chromosome 20 has been proposed, including (1) drug-resistant focal dyscognitive seizures (often occurring nocturnally and originating in the frontal lobe), (2) recurrent non-convulsive status epilepticus, and (3) classic EEG patterns. In ring chromosome 20, bifrontal slow waves, brief frontal epileptic discharges and long-lasting high-voltage slow waves with occasional uni- or bilateral frontal spikes, and/or frequent trains of theta waves in frontotemporal areas that are not influenced by eye-opening can be key features found on EEG that should suggest consideration of ring chromosome 20. Other seizures types can include tonic seizures and generalized tonic-clonic seizures. Seizures are typically drug-resistant, although there are case reports of seizure reduction with lacosamide and ezogabine.
Development. Most individuals with ring chromosome 20 have some degree of learning difficulty or intellectual disability, although there is a wide spectrum. Many individuals experience an initial cognitive decline with seizure onset and can experience this in further periods of uncontrolled seizures and/or non-convulsive status epilepticus. Some individuals regain these lost skills, especially during periods of better seizure control. There are many confounding variables to consider, including complex epilepsy management regimens that are often sedating.
Behavior. Behavior and attention difficulties are common in individuals with ring chromosome 20, including hyperactivity, difficulty concentrating, irritability, and obsessive behaviors. Some individuals have features or a diagnosis of autism spectrum disorder.
Genotype
Ring chromosomes. When a copy of a chromosome has formed a ring rather than the typical linear chromosome structure, this is called a ring chromosome. Ring chromosomes are not unique to chromosome 20 – in fact, ring chromosomes have been detected in all human chromosomes, and they are not always associated with any clinical phenotype. Other ring chromosomes, such as ring chromosome 14, have been also associated with epilepsy.
Genotype vs. phenotype. Ring chromosome 20 often occurs in the mosaic state, meaning that some cell lines do not have ring chromosome 20. There is some evidence that level of mosaicism may correlate with various measures of disease severity, such as age of epilepsy onset.
Segregation. In most cases, ring chromosome 20 occurs sporadically. However, inheritance from mosaic parents with varying levels of affectedness have been reported. Ring chromosomes are unstable during cell division, and there are several hypotheses on the mechanism of transmission between generations. Typically, when inheritance from a mosaic parent occurs, the resulting individual also has mosaicism, usually at a higher level of mosaicism than their parent. Thus, it is important to consider parental karyotyping when diagnosing an individual with ring chromosome 20.
Genetic testing methods. The gold standard for the diagnosis of ring chromosome 20 is a karyotype. It is important to emphasize this because ring chromosome 20 cannot be reliably detected with exome or genome sequencing nor chromosomal microarray. Although some cases may be detected on chromosomal microarray, if there is no overall loss of genetic material, then chromosomal microarray can miss the diagnosis.
Mechanism
Ring formation. A ring chromosome will form from the fusion of a chromosome’s polar ends, which is possible in multiple settings. These include (1) failed repair from a double-stranded break creating the possibility for subsequent fusion of the chromosomal poles. In this case, it is possible for genetic material at either pole to be lost depending on the location of the double-stranded break. To our knowledge, there is no specific region within chromosome 20 that has a greater tendency towards these breaks and subsequent reunions. (2) A second proposed mechanism is junction of the telomeres as a result of critical shortening of telomeric repeats. In this case, it is presumed that there is minimal loss of genetic material. (3) The third mechanism for ring formation is believed to be the product of a complex rearrangement (inversion/deletion/duplication rearrangements) leading to the fusion of arms. In this case there is the possibility for loss or gain of genetic data, though this would likely correlate with the details of the rearrangement itself.
Ring to phenotype. The nature of clinical features associated with ring chromosome 20 are still being characterized. Genes believed to be related to phenotypic features of ring chromosome 20 include CHRNA4, KCNQ2, DNAJC5, though individuals with ring chromosome 20 have been reported to have epilepsy without loss of these loci suggesting mechanisms beyond haploinsufficiency. Moreover, the epilepsy phenotype seen does not align with any of the typical presentations in these genetic conditions, suggesting that there is more to the story. Other theories related to phenotypic mechanism include epigenetic silencing of genes proximal to the telomere, variations of gene expression as a result of ring formation, and ring formation leading to greater cellular instability. Of note, it has been reported that a smaller ring may correlate with increased stability—perhaps due to a lower probability for crossover events to occur during meiosis which may lead to further chaos in the chromosomal architecture.
Community
The Ring 20 Research and Support network is a non-profit organization for families with ring chromosome 20 which provides support and community as well as ongoing research efforts and resources for clinicians.
References
Atkins, L et al. “A ring-20 chromosome.” Journal of medical genetics vol. 9,3 (1972): 377-80. doi:10.1136/jmg.9.3.377
Conlin, L K et al. “Molecular analysis of ring chromosome 20 syndrome reveals two distinct groups of patients.” Journal of medical genetics vol. 48,1 (2011): 1-9. doi:10.1136/jmg.2010.080382
Gago-Veiga, A B et al. “Specificity of electroclinical features in the diagnosis of ring chromosome 20.” Epilepsy & behavior : E&B vol. 80 (2018): 215-220. doi:10.1016/j.yebeh.2017.12.001
Onder, H and Tezer, F I. “Significant Improvements of EEG and Clinical Findings With Oral Lacosamide in a Patient With Ring Chromosome 20.” Clinical EEG and neuroscience vol. 47,4 (2016): 330-332. doi:10.1177/1550059415593428
Peron A, Catusi I, Recalcati MP, et al. Ring Chromosome 20 Syndrome: Genetics, Clinical Characteristics, and Overlapping Phenotypes. Front Neurol. 2020;11:613035. Published 2020 Dec 8. doi:10.3389/fneur.2020.613035
Tayama, Takahiro et al. “Improvement of epilepsy with lacosamide in a patient with ring chromosome 20 syndrome.” Brain & development vol. 42,6 (2020): 473-476. doi:10.1016/j.braindev.2020.03.003
Walleigh, D J et al. “Ring chromosome 20: a pediatric potassium channelopathy responsive to treatment with ezogabine.” Pediatric neurology vol. 49,5 (2013): 368-9. doi:10.1016/j.pediatrneurol.2013.06.005

